While West Africa faces the ravages of Ebola, potential vaccines developed a decade ago are being exhumed from what Dr. James E. Crowe calls “the biotech valley of death.” Dr. Crowe, Director of the Vanderbilt Vaccine Center at Vanderbilt University, describes the valley as the tenuous transition researchers face in moving drugs from animal to human trials. At this point, many potentially life-saving medical technologies and pharmaceuticals have been relegated to laboratory shelves. In 2000, a group of scientists based out of the Centers for Disease Control and Prevention (CDC) and the National Institutes of Health’s (NIH) Vaccine Research Center published a paper in Nature Journal titled “Development of a preventive vaccine for Ebola virus infection in primates.” Three years later, a second group of American scientists built on this study in another paper in Nature describing the development of a primate-tested vaccine that “could provide an intervention to limit the epidemic spread of Ebola.” Despite the vaccine’s apparent potential, however, trials never occurred. Now, as the world faces the largest Ebola epidemic in history — with the CDC estimating a worst-case scenario of 1.4 million cases of the disease by January 2015 — scientists, politicians, health organizations and the distraught public are belatedly rushing these vaccines to clinical trials, leaving a string of ethical questions in their wake.
The World Health Organization (WHO) has sprinted to the forefront of this fight; the organization convened in October to discuss the framework for and planning of clinical trials. Volunteer-based trials to test the safety of the vaccine began in the United States in October, and similar trials are scheduled to take place in Canada, Britain, Switzerland, Germany, Mali, Gabon and Kenya. Two significantly larger trials are set to start as early as January in Liberia and Sierra Leone, two nations that have borne the brunt of the epidemic. But, according to the WHO official Marie-Paule Kieny, the world’s mobilization to rush vaccinations to clinical trials, “accelerating in a matter of weeks a process that typically takes years,” has raised a number of ethical quandaries and logistical problems. With the growing complexity of global health infrastructure and clinical trials, the media and the public have asked important questions regarding the morality of placebo-based trials, the selection of target populations and the infrastructural difficulties of initiating trials. While these are critical inquiries, the Ebola crisis has also uncovered even larger issues about why medical technologies do not go to trial when they should. By uncovering these shortcomings, the Ebola epidemic has provided the global health establishment more than just an opportunity to critically reevaluate its approach to and incentives for clinical research and trials — it is an opportunity to effect structural, long-lasting change.
Perhaps the most significant problem within the biomedical research field that the Ebola epidemic has unveiled is the underinvestment in research and development in the infectious disease sector. This is partly responsible for the fact that early-stage vaccinations for Ebola, developed over a decade ago, sat on the backs of laboratory shelves until now, after over 5,000 individuals have already lost their lives to the disease. Dr. Jonathan Kimmelman, Associate Professor of Biomedical Ethics at McGill University, explained to the Brown Political Review: “The ethical issue is not who is going to get access to these drugs that are in Phase I or Phase II tests. The ethical issue here is why, as societies, we underinvest in addressing major infectious-disease health threats.”
Ebola and many other infectious tropical diseases fall victim to the “orphaned research” phenomenon. Even promising drugs and vaccines are often orphaned in their nascent stage. According to the Food and Drug Administration, such “drugs and biologics are intended for safe and effective treatment…but are not expected to recover the costs of developing and marketing,” either because they affect only a small subset of the population, or because they provide little opportunity for substantial profit over high development costs. The prohibitive price tags associated with developing preventative technologies (such as vaccinations), as well as cures for diseases that, like Ebola, predominantly affect less wealthy equatorial countries, have helped engender a category of infectious diseases referred to as “neglected tropical diseases” (NTD). Like Ebola, the 17 other NTDs, which include rabies and dengue fever, have fallen victim to a system that does little to create or sustain investment in high-cost, low-return vaccines.
Obscured behind vague medical terminology and funding figures are the human lives affected by orphaned research and the associated underinvestment in treatments and preventative technologies: NTDs kill an estimated 534,000 of the poorest one billion people on the planet every year. Because the diseases have little to no impact in the developed world, there is little economic incentive for drug companies to invest in research for preventative and treatment technologies. Consequently, progress against these diseases proceeds at a slow pace and is largely contingent on philanthropic and government funding.
A 2009 analysis commissioned by the Bill and Melinda Gates Foundation found that in 2007, $2.5 billion was invested globally in research and development for new “neglected disease products.” Of this money, 90 percent, or $2.3 billion, was given by public donors (such as the NIH and other government-funded groups) and private philanthropists. Reflecting the low profitability of this research area, the pharmaceutical industry provided only $231.8 million — 9 percent of global investment — in research and development for NTDs. Furthermore, not all NTDs receive equal attention: The report found that “funding was highly concentrated, with HIV/AIDS, tuberculosis, and malaria receiving nearly 80 percent of the total.” Notably, all three of these diseases, while still more prominent in less developed regions of the world, are much more prevalent in the developed world than are other NTDs.
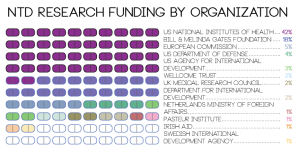 Despite the large amount of private capital available to for-profit pharmaceutical companies that command the global medical research and development sector, few of these resources are put towards unprofitable NTD vaccines. In the United States — the largest global investor in health research and development — approximately 54 percent of health research and development funding is provided by private pharmaceutical corporations. The WHO reports that “even in the United Kingdom, where the medical profession receives more independent, publicly-funded information than in many other countries, promotional spending by pharmaceuticals companies is 50 times greater than spending on public information on health.” Because large for-profit corporations carry out the majority of research and development globally, the pursuit of new medical technologies is inherently influenced by their perceived future profitability.
Despite the large amount of private capital available to for-profit pharmaceutical companies that command the global medical research and development sector, few of these resources are put towards unprofitable NTD vaccines. In the United States — the largest global investor in health research and development — approximately 54 percent of health research and development funding is provided by private pharmaceutical corporations. The WHO reports that “even in the United Kingdom, where the medical profession receives more independent, publicly-funded information than in many other countries, promotional spending by pharmaceuticals companies is 50 times greater than spending on public information on health.” Because large for-profit corporations carry out the majority of research and development globally, the pursuit of new medical technologies is inherently influenced by their perceived future profitability.
It was this phenomenon that kept the 2000 and 2003 Ebola vaccines shelved. Thomas Geisbert, one of the developers of the 2003 vaccine and a researcher at the University of Texas Medical School’s Galveston branch, told the New York Times that “there’s never been a big market for Ebola vaccines. So big pharma, who are they going to sell it to?” Vaccines simply are not profitable for pharmaceutical companies. Even though the value of the vaccine market increased from $5 billion to $24 billion between 2000 and 2013, this represents merely 2.5 percent of the pharmaceutical industry’s $955 billion revenue in 2013. On balance sheets in which revenue reaches almost one trillion dollars, a few billion is little more than a drop in the pond. The WHO recognizes that this conflict of interest can mean difficulties in getting important drugs and vaccines developed, acknowledging that “the profit imperative ensures that the drugs chosen for development are those most likely to provide a high return on the company’s investment. As a result, drugs for use in the industrialized world are prioritized over ones for use in the [developing world], where many patients would be unable to pay for them.” This problem — with no easy fix — contributed significantly to the failure to pursue further trials for promising Ebola vaccines.
The WHO estimates that the development of vaccines can cost anywhere from $163 million to $518 million and can take 15 or more years. While wealthy Western governments have the means to subsidize research and development in new medical technologies that might have enormous social benefit but little profit potential, governments in the developing world don’t have the same financial capacity. Because neither poor countries nor their citizens can afford to bankroll medical research and development for “unprofitable” diseases, there are even fewer incentives for pharmaceutical companies to pursue vaccines or treatments for diseases primarily affecting the developing world. Consequently, the developing world is left largely dependent on Western governments and agenda-driven private philanthropy to underwrite research and development for diseases that disproportionately affect it.
This is not to say that Western countries shouldn’t take up these causes — just that they rarely do so to the necessary extent. After all, Ebola has shown that developed nations can’t simply rest easy — no country is entirely immune to the threat of epidemics. But the sudden interest amongst pharmaceutical companies to begin clinical trials on an Ebola vaccine is not the product of simple altruism. The Norwegian Institute of Public Health has estimated that as many as 10 to 27 million doses of a vaccine are needed due to the recent crisis — numbers which have surely piqued pharmaceutical interest. And with Western governments increasingly worried about their own populations’ chances of exposure, the profitability of medicines to combat and prevent Ebola has increased. Johnson & Johnson, one pharmaceutical company working on developing a vaccine, has already invested a total of $387 million in a potential solution — $200 million to accelerate the production of the first of a two-part vaccine and $187 million to acquire the rights to the second part of the vaccine from a Danish company.
While it is easy to blame “Big Pharma” for previously failing to bankroll research into medical technologies and pharmaceuticals with the potential to eradicate and treat diseases like Ebola, they may be taking more flak than is due. Large pharmaceutical companies have the capability to maintain capital, technology and personnel that government institutions don’t — resources that have proven immensely valuable — in part because of the profitability of their products. Since the system is unlikely to change outright, government policies that induce private interests to take up research on diseases like Ebola and other NTDs might be a constructive step forward for both policymakers and the pharmaceutical industry. The current Ebola crisis can serve as the catalyst for this type of change. The epidemic has pulled the curtain aside on underinvestment in research and development for the types of infectious diseases that disproportionately affect the bottom billion. In doing so, it has created an opportunity to evaluate and change both public and private biomedical technology development policies across the globe.
Speaking with BPR, Dr. Jennifer Blumenthal-Barby, an assistant professor of Medicine and Medical Ethics at Baylor College of Medicine, suggested that “using the powers of social norms, ego and re-aligning incentives might be promising ‘nudges’ to use with drug companies.” The Orphan Drug Act of 1983 is one example of a policy nudge that can be used to induce pharmaceutical companies to rescue orphaned research and invest in less profitable areas of research and development. The act provides seven-year market exclusivity to “sponsors of approved orphan products” and “a tax credit of 50 percent [of] the cost of conducting human clinical testing.” The FDA reports that the program “has successfully enabled the development and marketing of more than 400 drugs and biologic products for rare diseases since 1983. In contrast, fewer than 10 such products supported by industry came to market between 1973 and 1983.” Furthermore, Dr. Blumenthal-Barby explained, “the ethically favorable thing about nudges (as opposed to mandates) is that they preserve the liberty of corporations or individuals since they are just that — nudges — and these entities may still choose a different option.” In light of the neglect of NTDs by the world of corporate-dominated medical research and development, leading countries like the United States should consider implementing nudging laws that apply directly to infectious diseases such as Ebola.
The class-specific barriers to drug development are not the only deep-seated issues that the Ebola epidemic has unearthed. The acceleration of the clinical trial process has also exposed fundamental flaws in the way we currently approach and conduct clinical trials themselves. Western media has mostly focused on the ethical concerns of using placebo-based control groups in the trials that are set to occur in West Africa (for instance the upcoming trial in Liberia). While accepting the usefulness of placebos in other cases, experts like David Heymann, the head of the Chatham House Center on Global Health Security, have said that the use of placebos can’t be justified “in the midst of the worst Ebola epidemic in history.” Although the concerns of experts like Heymann are understandable, the use of the placebo in this case is not the main ethical concern. As explained by Dr. Kimmelman, a well-designed clinical trial utilizes the concept of clinical equipoise — the understanding that a trial should function only so long as it can work under a null hypothesis. In other words, throughout the length of the trial there should be no reason to believe that any group of test subjects, regardless of whether they receive the placebo or not, will be any worse off than they would be otherwise. “In the case of Ebola,” says Dr. Kimmelman, because the standard of care is essentially nonexistent, “giving people a placebo is not inconsistent with the standard of care, so it’s not unethical; you’re not putting people at greater risk in order to test something.”
While this sentiment may still leave many uncomfortable, anti-placebo concerns are predicated on mistaken assumptions. The overwhelming concern with the morality of placebos relies on a general belief that those receiving the placebo will inherently be worse off than those who don’t receive it. This assumption points to misplaced optimism as to the efficacy of experimental drugs and vaccines and to a general lack of public knowledge about the problems that clinical trials face. “In the media-portrayal of the [Ebola] vaccine studies, there has been a presumption that this vaccine will ultimately prove to be useful. But the reality is that the vast majority of drugs, vaccines and other interventions that make it into clinical development end up either being unsafe or being ineffective, or both,” explains Dr. Kimmelman. The rushed pace of the Ebola clinical trials in particular should make the public even more wary of placing too much stock in the vaccine’s success.
Global efforts to develop vaccines to protect against malaria and HIV, for example, have been largely in vain. Unlike Ebola, malaria is an ever-present problem in tropical regions, killing an estimated 627,000 people per year, and as such it has received significantly more attention from the medical research world than Ebola has. Despite the fact that scientists have been studying and attempting to defeat malaria for decades, no vaccine has yet proven successful. The WHO reports that although over 20 malaria vaccine variants are currently in clinical trials, only one vaccine in the history of malaria vaccine research and development has made it to Phase III clinical trials (in which testing begins in large test-groups; Phase I and II trials also use human subjects but smaller test-groups). Although the malaria vaccine currently in development looks more promising than its predecessors, it will not be ready for some time and, thus far, has only demonstrated 51 percent efficacy. Similarly, the search for a vaccine to protect humans against HIV has been a long and fruitless battle. The first clinical trial for a potential HIV vaccine began in 1987, but 30 vaccine trials and 27 years later, the WHO reports that “only two related [HIV] candidate vaccines [are] being evaluated in Phase III efficacy trials.”
Uneducated optimism surrounding novel vaccines may lead us to disregard the very real risks faced by the test subjects of new vaccines, such as those developed for Ebola. Novel vaccines, explains Dr. Kimmelman, have real potential to “make people worse off.” In 2008, it was discovered that a promising HIV vaccine — the STEP vaccine — which had begun Phase II trials in 2003, may have actually made participants more vulnerable to contracting HIV. Test subjects who had received the vaccine were contracting HIV at a higher rate than those who had not. While the vaccine almost certainly did not cause HIV to develop, its role in the increased incidence of HIV in vaccinated participants remains unclear, and the STEP trials serve as a cautionary tale when it comes to the general optimism that surrounds the rollout of experimental vaccines.
The immense complexities behind conducting clinical trials are partly responsible for the high rate of failure when medical technologies, such as vaccines, make the leap from non-human to human trials. But the greatest problems begin before new medical technologies even reach clinical trials. Dr. Kimmelman describes the current state of preclinical research as “a mess” with “so many problems and distortions in the way we do preclinical work that it’s hard to know what you can trust and what you can’t.” Preclinical trials are subject to publication bias and high incidences of type I errors, also known as false-positives. This is because clinical trials often aren’t subject to the most stringent testing standards, like randomization, physician and patient blinding and large sample sizes. Additionally, because publications have a tendency to only publish positive findings, it is hard for researchers to contextualize and interpret successful trials and to distinguish between accurate and inaccurate results.
In light of the Ebola epidemic, reforms to enhance the preclinical trial process are all the more pressing, as the faith put into Ebola vaccines is largely rooted in their perceived success in such trials. But given the tendency for successful preclinical trials to fail once they reach human trials, the celebration may be coming too soon. Since failure due to poor preclinical work can result in hundreds of millions of dollars lost and countless resources squandered in the midst of a time-sensitive epidemic, holding trials to higher research standards increases chances of success in times of crisis.
All this does not go to say that there’s no merit in conducting clinical trials for the current Ebola vaccinations. For one, clinical trials depend on the existence of an at-risk population, which, unfortunately, the current epidemic has provided. While circumstances will certainly complicate the clinical trials, the ability to test a vaccine today may help save thousands of lives from Ebola tomorrow. However, the public must remain aware that vaccinations are not infallible, and given the poor state of the preclinical research realm, the global community should be wary of putting all its eggs in one basket.
The Ebola epidemic has opened the door to questions that the medical world has long avoided asking, but to which the answers would be invaluable. The ability to refine response mechanisms in the future will largely depend on the willingness to reexamine and take steps to improve the current system of pharmaceutical research and development. For the first time in decades, the Western world has tasted some of the fear that citizens of the global south face daily, and while the Ebola epidemic will eventually pass, the looming specter of disease will not disappear. After a summer of death in West Africa, it’s no wonder that the emergence of a possible vaccine to protect against Ebola has ignited the public’s imagination. But even more valuable than the vaccine itself could be the lessons that we draw from it.
Art by Annabel Ryu.
